Primary Pharmaceuticals

About Us
Primary Pharmaceuticals is a privately held, specialty pharmaceutical distributor with the goal of providing its network of wholesalers and independent pharmacies innovative, valued and quality products at a low out-of-pocket cost to their customers.
Primary’s line of niche pharmaceutical products are developed, produced and marketed in the USA through a diverse partnership of industry leading designers, packagers and distributors from across the country. Primary’s products are registered with the FDA and listed with the top pharmaceutical databases like First Databank, Medispan and Redbook.
We pride ourselves in providing our clients with quality products manufactured to pharmaceutical standards using the latest cGMP guidelines to ensure our customers received a product which not only meets, but surpasses industry requirements.
Our Products
We guarantee quick access to a wide range of leading pharmaceuticals, including supply-sensitive products.
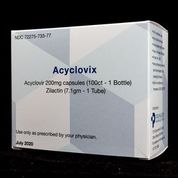
Acyclovix
NDC# 72275-0733-77
Call for details
AWP $1,890
1 x Acyclovir 200mg 100CT & 1 x Zilactin
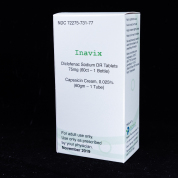
Inavix
NDC# 72275-0731-77
Call for details
AWP $1,955.00
1x Diclofenac Sodium DR 75mg Tablet 60ct & 1x Capsaicin 0.025% Cream 60gm
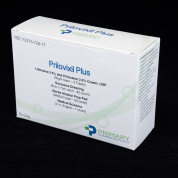
Prilovixil Plus
NDC# 72275-0726-77
Call for details
AWP $$2,900
3 x Lidocaine 2.5%/ Prilocaine 2.5% 30g & 20 x Occlusive Dressings & 20 x Alochol Prep Pads & 1 x Medical Scissors

